 
When drawing Lewis dot structures, the overall charge on a polyatomic ion is equal to the sum of the formal charges on each atom in the ion. This is to show that the subscript applies to the entire polyatomic ion. In a polyatomic ion, the group of covalently bonded atoms carries a net charge because the total number of electrons in the molecule is not equal to the total number of protons in the molecule. Oxyanions that contain elements in the same group of the periodic table have an analogous formula and are named in a similar convention. Any such molecule that has nonzero net charge is a polyatomic ion. 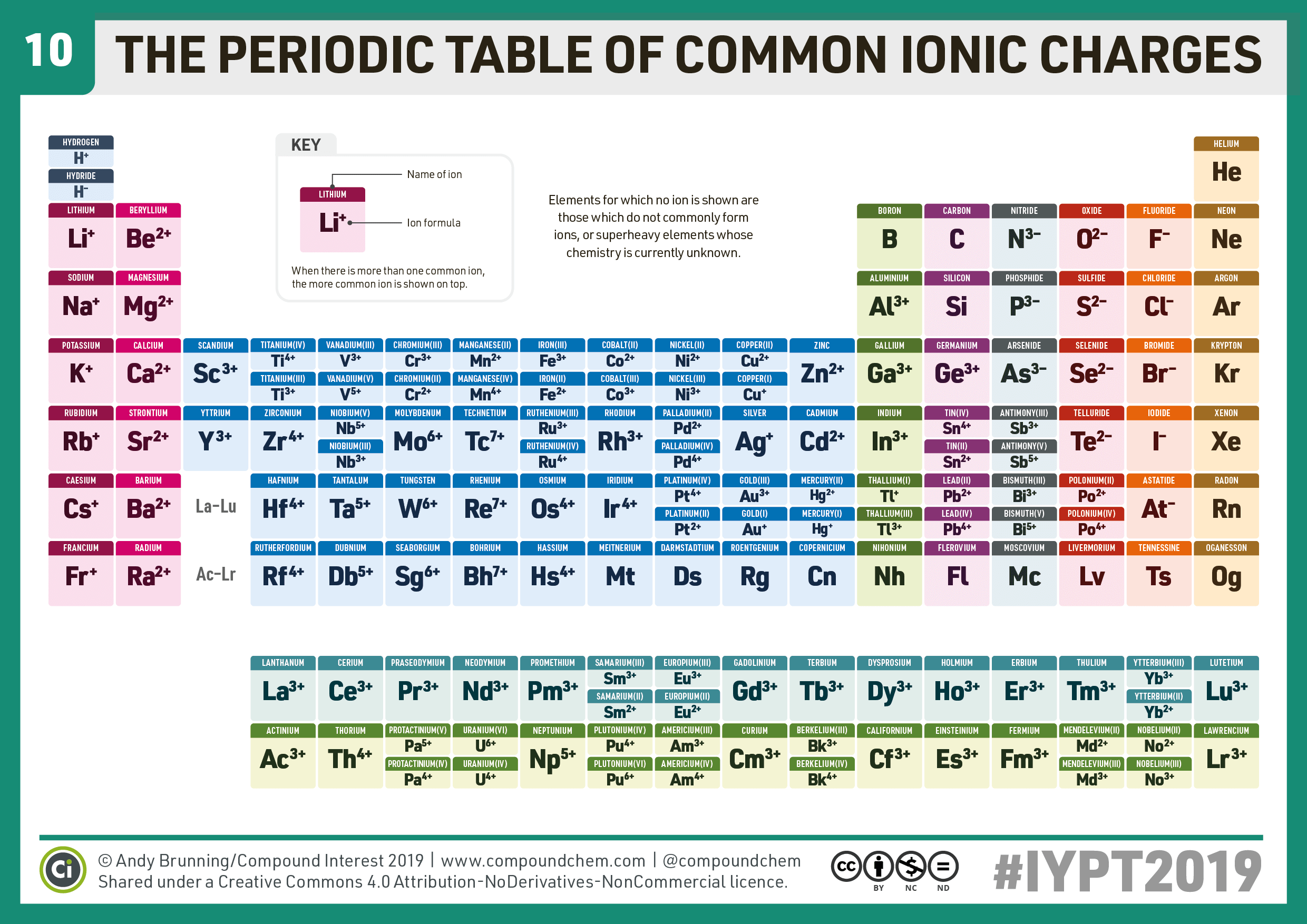
If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. 3.2 Ions and the Periodic Table Common Cations Common Anions Ions of Transition Metals 3.3 Ionic Bonding 3.4 Practice Writing Correct Ionic Formulas 3.5 Naming Ions and Ionic Compounds 3.6 Polyatomic Ions 3.7 Naming Polyatomic Ions 3.8 Properties and Types of Ionic Compounds 3.9 Arrhenius Acids and Bases 3. protons electrons So, hydroxide has a net charge of 1 because its number of electrons exceeds its number of protons by one. It has five atoms (one nitrogen and four hydrogens) that share a charge of. The carbonate ion consists of one carbon atom and three oxygen atoms, and carries an overall charge of 2. Polyatomic ions are charged groups of atoms. 
Together, they comprise a single ion with a 1 + charge and a formula of NH + 4. The ammonium ion consists of one nitrogen atom and four hydrogen atoms. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. A polyatomic ion is an ion composed of more than one atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
